To make a material that is colored, one normally uses a dye or pigment. But another way to make color is to make a nanostructure that reflects or scatters light so that waves of certain frequencies can constructively interfere. These nanostructured materials are said to have structural color. Unlike traditional color, which comes from dyes or pigments that absorb light, structural color can be made resistant to fading. We use colloidal self-assembly to make nanostructures that have a variety of different colors. At the same time, we aim to understand the physics of the scattering process so that we can optimize these nanostructures for applications.
We are particularly interested in making structural color that is angle independent. That means that the color is the same, regardless of how you rotate the material, and regardless of the angle between the light source and your eyes. There are many structurally colored materials that, like an opal stone, are iridescent, meaning that the color changes with the viewing angle and orientation. The reason for this change in color is that the nanostructure of these materials is well-ordered (or crystalline). Sometimes these kinds of ordered structures are called photonic crystals.
To make materials that have angle-independent color, we need to make nanostructures that are disordered. We call these materials photonic glasses (a term that we believe was first coined by John Ballato in 2000). Examples of naturally occurring photonic glasses are the feathers of blue birds and the wings of blue butterflies. Eric Dufresne and collaborators were the first to demonstrate synthetic structures that mimic the blue color of the bird feathers (Forster, Noh, Liew, Saranathan, Schreck, Yang, Park, Prum, Mochrie, O'Hern, Cao, Dufresne Advanced Materials 2010). They made these structures from spherical colloidal particles, and they taught us their techniques.
Our goal is to study, both theoretically and experimentally, how the optical properties of these glasses relate to their structure and constituent particles. Based on our observations from photonic glasses of conventional particles, we constructed a theoretical model that explains why it is difficult to make yellow, orange, and red photonic glasses. Guided by this model, we have developed new colloidal systems that give a higher degree of control over structural color. These systems might be used to make reflective displays or paints and coatings.
Understanding why blue structural colors are easier to make than red
Early in our work on angle-independent structural color, we found that it was easy to make blue colors but hard to make yellow, orange, or red. It also turns out that nearly all examples of angle-independent color in nature are blue. Sofia Magkiriadou and Jin-Gyu Park set out to understand why. Sofia made photonic glasses of different particle sizes and showed that the wavelength of the structural color scaled with the size of the particle, as expected. She was able to make blue and green samples, but the sample that should have been red turned out to be magenta:
 At left, measured reflectivity spectra for three similarly prepared colloidal glasses of PMMA particles in air. Photographs of the samples are shown at right. The magenta sample would appear red if it weren't for the high reflectivity in the blue, indicated by the arrow in the spectra.
At left, measured reflectivity spectra for three similarly prepared colloidal glasses of PMMA particles in air. Photographs of the samples are shown at right. The magenta sample would appear red if it weren't for the high reflectivity in the blue, indicated by the arrow in the spectra.
Sofia developed a theoretical model to explain the blue peak in the reflectivity spectrum that, when mixed with the red structural color, made these samples magenta. She showed that this blue peak arises because the individual colloidal particles tend to scatter light more strongly in the blue than in the red (the same argument can be used to explain why the sky is blue). More technically, she and her colleagues showed how the backscattering resonances of the individual particles arise from cavity modes and how these resonances interact with the constructive interference of the nanostructures to suppress the structural red color. From the model that Sofia developed, we were able to establish some design rules for making red structural color. Our findings were highlighted in Physics, New Scientist, and Chemistry World.
Making angle-independent red structural colors
Based on the design rules established in our theoretical study, our group is trying to make photonic pigments that are non-fading and non-toxic: these pigments can potentially be used in cosmetics, reflective displays, and inks.
Jin-Gyu Park, working together with Sofia and Shin-Hyun Kim, were able to demonstrate a colloidal assembly method to make microcapsules that showed non-iridescent structural colors spanning the entire visible range. The microcapsules are made using a microfluidic device that produces a droplet surrounded by a thin shell of monomer. The droplet contains colloidal particles with a core-shell structure, the core consisting of polystyrene, which has a high-refractive index, and the shell consisting of a hydrogel, which has a low refractive index. The core-shell structures of the particles allows us to decouple the distance between the particles, which sets the structural color, from the scattering strength of the individual particles, which sets the opacity (to understand this, have a look at one of our earlier papers).
After making the droplets, Jin-Gyu and colleagues were able to concentrate the core-shell particles into a dense, disordered nanostructure by pulling out the water. Then they polymerized the shell to make photonic capsules:
 Top photographs show a droplet containing core-shell scatterers. By removing the water, as shown in the diagram at bottom, we concentrate the particles. The color of the droplet becomes more yellow as the particles get more concentrated. We can then lock in the structure by polymerizing the shell, creating a photonic capsule. The electron microscope image at bottom right shows the random and isotropic nanostructure formed by the core-shell particles inside the capsule.
Top photographs show a droplet containing core-shell scatterers. By removing the water, as shown in the diagram at bottom, we concentrate the particles. The color of the droplet becomes more yellow as the particles get more concentrated. We can then lock in the structure by polymerizing the shell, creating a photonic capsule. The electron microscope image at bottom right shows the random and isotropic nanostructure formed by the core-shell particles inside the capsule.
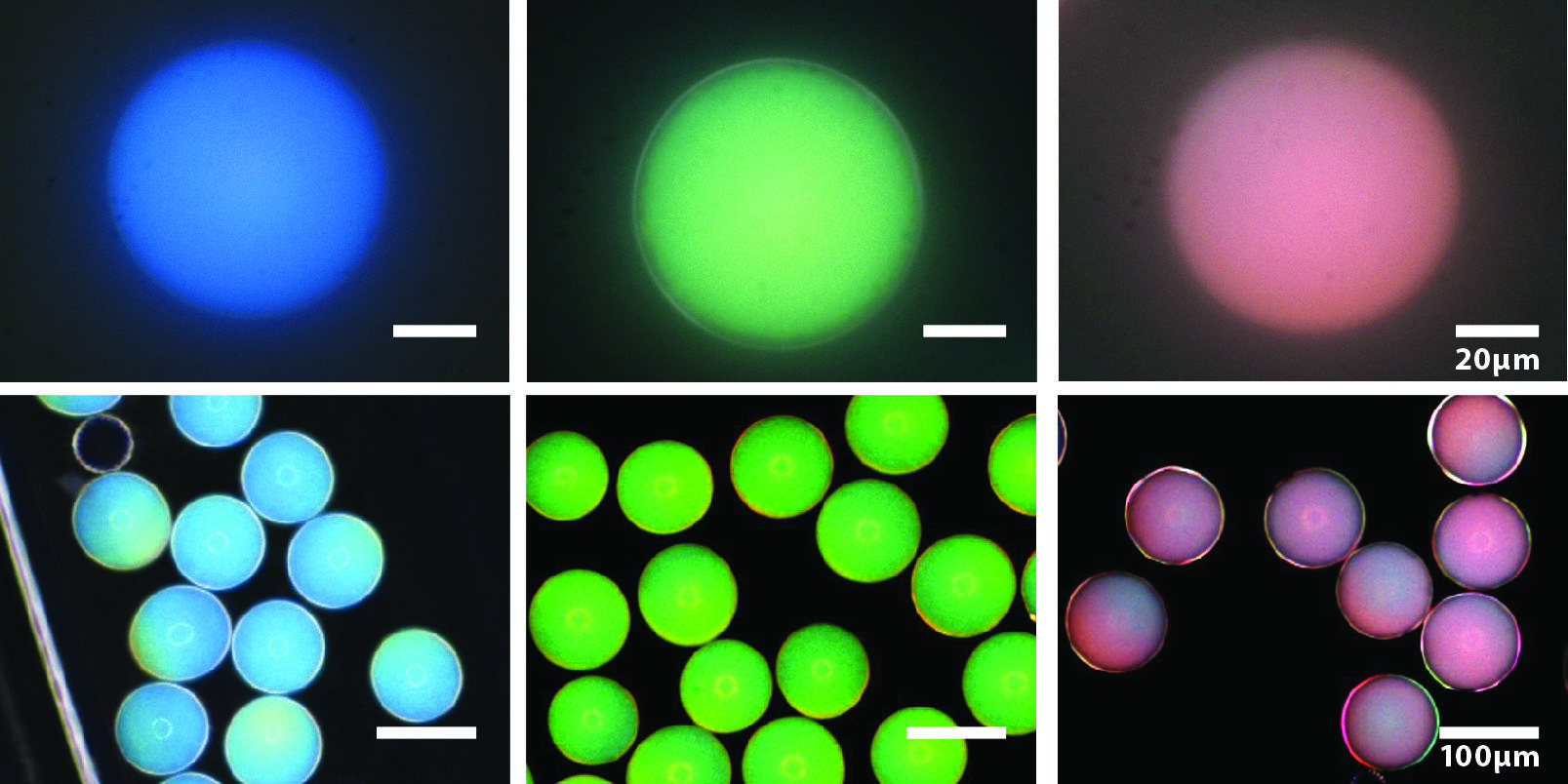
By changing the sizes of the cores and the shells, Jin-Gyu and colleagues were able to make photonic capsules with blue, green, and red colors:

Our results were highlighted in Phys.org, Chemical & Engineering News, Harvard Magazine, and in a press release from Harvard SEAS.
Materials with responsive structural color
Jin-Gyu Park and Ben Rogers realized that the hydrogel core-shell particles we were using to make the photonic capsules could also be used to make responsive structural color, that is, color that can change with solution conditions or temperature. They made the photonic crystals shown below, which can shift their color rapidly in response to temperature. These materials might be used as sensors or as resonators for organic lasers.
 Colloidal photonic crystals made of hydrogel building blocks can rapidly change colors without melting
Colloidal photonic crystals made of hydrogel building blocks can rapidly change colors without melting





